The AP Chemistry exam is a challenging high school science assessment, and the free-response section plays a major role in determining a student’s final score. Unlike multiple-choice questions, FRQs require students to communicate their thinking through calculations, explanations, and experimental reasoning, making approach and clarity essential for earning full credit.
If you’re still deciding whether AP Chemistry is the right fit for you, our AP course selection article can help you choose courses that align with your academic goals.
This guide breaks down AP Chemistry FRQs, offers practical strategies, and reviews sample questions so you understand what is required.
What Is the AP Chemistry FRQ Section Format?
The free-response section evaluates skills such as modeling, representation, experimental design, method selection, data interpretation, quantitative problem solving, and scientific argumentation.
Key features of the free-response section include:
- 105 minutes total (1 hour and 45 minutes)
- Calculator use is permitted
- Seven free-response questions in total
- Three long-response questions (10 points each)
- Four short-response questions (4 points each)
As a general time guide, aim to spend around 20 minutes on each long-response question and roughly 9 minutes on each short-response question. This gives you a little extra time to review your answers before time is up.
With the format clear, the next section offers a step-by-step approach to AP Chemistry FRQs.
How to Answer AP Chemistry Free-Response Questions
Answering AP Chemistry FRQs well requires structure, clarity, and alignment with scoring guidelines. Points are awarded for clear reasoning, not just correct answers, so how you show your thinking matters as much as the result. The following steps provide a structured approach for answering free-response questions:
1. Read the question carefully before writing anything
Understand each part before answering. Identify if the particular question requires calculation, explanation, data analysis, or experimental reasoning. Misreading the given question is a common and preventable reason for losing points.
2. Address each part separately
Each FRQ part is scored separately, so answer every section, even if unsure. Do not combine responses, and if a later part depends on an earlier answer, explain your logic throughout. Partial credit is common for clear reasoning; never leave sections blank.
3. Show every step of your reasoning
AP Chem FRQs reward explanations, not just correct answers. Clearly show calculations with units, explain your reasoning using chemical principles, and reference any data, equations, or trends from the question.
4. Use precise scientific language
Be clear and specific in your response. Use proper terminology, avoid vague statements like “it increases” without context, and clearly state assumptions when asked. Precision is essential, especially in error analysis, where you must explain specific causes and impacts.
5. Review for completeness before moving on
If time allows, take a moment to confirm that each part of the question has been addressed, check calculations for unit consistency, and ensure your explanations directly respond to the question. A brief review can prevent simple oversights that cost meaningful points.
AP Chemistry FRQ Examples (Long & Short Response)
The AP Chemistry exam is tough, and the free-response section is crucial for scoring high. Memorizing formulas isn’t enough; you need to clearly explain your reasoning and structure your answers to avoid losing points. Let’s explore this with examples and address common student questions.
Example 1: Long-Response FRQ (10 points)
Let’s take one of the key questions adapted from College board’s released AP Chemistry exam questionnaire. Students were given a long question centered around magnesium. It moved from isotopes to Coulomb’s law to solubility equilibrium, which makes it a great example of how layered the format of the AP Chemistry free-response questions really is.
We will now solve it step by step while also understanding how the AP Chemistry FRQ is graded along the way.
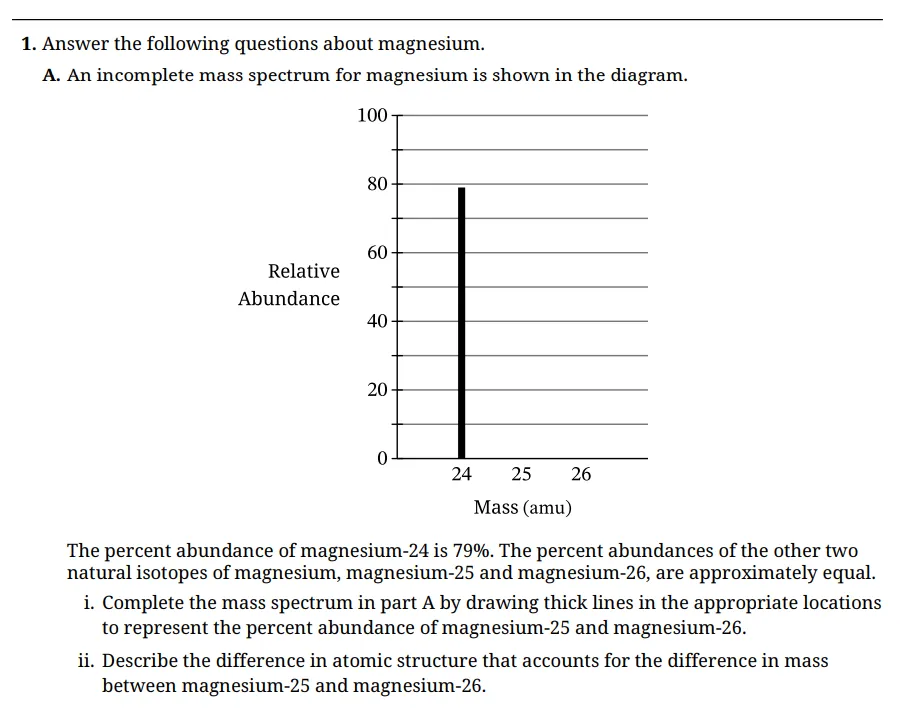
A. Mass Spectrometry (Isotopes)
We are told:
- Magnesium-24 has 79 percent abundance
- Magnesium-25 and Magnesium-26 are approximately equal
(All isotope abundances must add up to 100 percent)
100 − 79 = 21
Since Mg-25 and Mg-26 are equal: 21 ÷ 2 = 10.5 percent
(i) So the completed mass spectrum should show:
- 24 amu → 79 percent
- 25 amu → about 10 to 11 percent
- 26 amu → about 10 to 11 percent
Scoring insight: This earns 1 point if both remaining isotopes are shown at approximately equal heights. The graders are checking proportional reasoning, not artistic precision.
(ii) Why do Mg-25 and Mg-26 have different masses?
Magnesium always has 12 protons.
Mg-25 has 13 neutrons
Mg-26 has 14 neutrons
They differ in the number of neutrons, which explains the difference in mass.
Scoring insight: This earns 1 point only if the response clearly states a different number of neutrons. Answers like different masses or different structures are too vague. In an AP Chemistry FRQ, precise language earns credit.
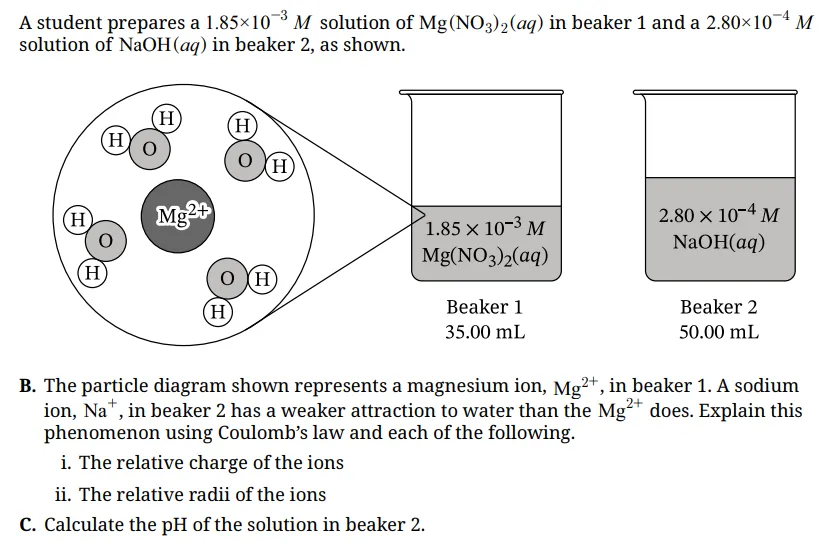
B. Coulomb’s Law
Why does Mg²⁺ attract water more strongly than Na⁺?
Coulomb’s law tells us that electrostatic attraction increases when charge increases and when the distance between charges decreases.
(i) Mg²⁺ has a plus 2 charge, while Na⁺ has a plus 1 charge
(ii) Mg²⁺ is also smaller than Na⁺, meaning its positive charge is more concentrated
Because Mg²⁺ has both a higher charge and a smaller ionic radius, it attracts the partially negative oxygen atom in water more strongly.
Scoring insight: This section is worth 2 points
- One point is earned for discussing higher charges
- One point is earned for discussing a smaller radius
If only one factor is mentioned, only one point is awarded. Each idea is scored independently.
C. pH Calculation
Given:
[OH⁻] = 2.80 × 10⁻⁴ M
First calculate pOH:
pOH = −log(2.80 × 10⁻⁴), pOH ≈ 3.55
Then calculate pH:
pH = 14 − 3.55, pH ≈ 10.45
Final Answer: pH = 10.45
Scoring insight: This earns 1 point for correct setup and calculation. Even if rounding differs slightly, consistent mathematical reasoning can still earn credit. The AP Chemistry FRQ rubric rewards correct process, not just the final number.
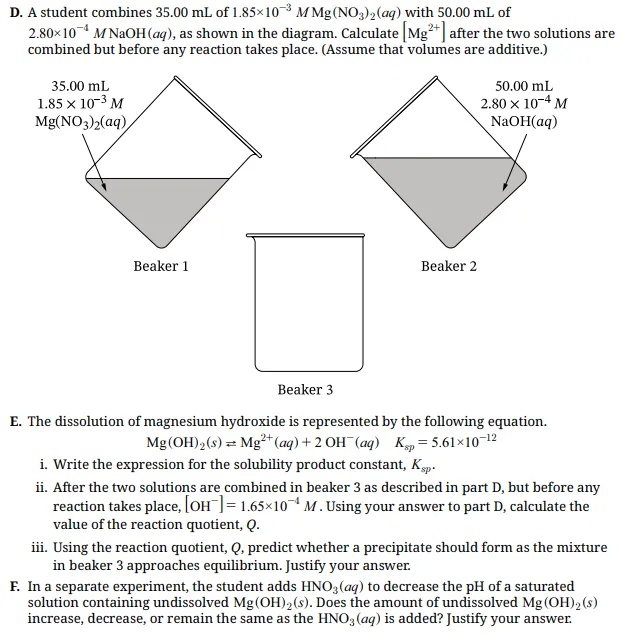
D. Concentration After Mixing (Before Reaction Occurs)
Step 1. Calculate moles of Mg²⁺
(1.85 × 10⁻³ M) × (0.03500 L) = 6.48 × 10⁻⁵ mol
Step 2. Determine total volume after mixing
35.00 mL + 50.00 mL = 85.00 mL = 0.08500 L
Step 3. Calculate new concentration
6.48 × 10⁻⁵ mol ÷ 0.08500 L = 7.62 × 10⁻⁴ M
Scoring insight: This earns 1 point for the correct dilution calculation. Even if arithmetic slips slightly, a correct setup often earns credit.
E. Ksp and Reaction Quotient
(i) Dissolution equation: Mg(OH)₂(s) ⇌ Mg²⁺ + 2OH⁻
Ksp expression: Ksp = [Mg²⁺][OH⁻]²
This earns 1 point for writing the correct Ksp expression.
(ii) Now calculate Q using:
[Mg²⁺] = 7.62 × 10⁻⁴ M
[OH⁻] = 1.65 × 10⁻⁴ M
Q = (7.62 × 10⁻⁴)(1.65 × 10⁻⁴)²
Q ≈ 2.07 × 10⁻¹¹
This earns 1 point for correct substitution and calculation.
(iii) Compare Q with Ksp:
Ksp = 5.61 × 10⁻¹²
Since Q is greater than Ksp, the solution is supersaturated and a precipitate will form.
Scoring insight: This earns 1 point only if the comparison between Q and Ksp is explicitly stated. Writing precipitate forms without showing Q > Ksp is not enough.
F. Adding HNO₃
Adding HNO₃ introduces H⁺ ions.
H⁺ reacts with OH⁻: H⁺ + OH⁻ → H₂O
This decreases the concentration of OH⁻. When OH⁻ decreases, equilibrium shifts to the right to replace it, causing more Mg(OH)₂ to dissolve.
Scoring insight: This earns 1 point if both ideas are clearly explained. The response must state that OH⁻ decreases and that equilibrium shifts to dissolve more solid.
What This Shows About AP Chemistry FRQ Grading
This single long question was worth about 10 points total, divided across small scoring components.
Each concept earns its own point:
- Isotope reasoning
- Electrostatic reasoning
- pH calculation
- Dilution math
- Ksp expression
- Q comparison
- Le Châtelier explanation
Key takeaway: You could miss one calculation and still score well because points are distributed across ideas. That is how long-response questions are designed. The exam rewards structured reasoning, not perfection.
Example 2: Short-Response FRQ (4 points)
Here is a Short-Answer Question 4 adapted from College board’s released AP Chemistry questionnaire. This problem brings together intermolecular forces, hybridization, phase changes, and enthalpy of vaporization, making it a great example of how the format of the AP Chemistry free-response questions combines conceptual understanding with calculation. Let’s work through it step by step and see exactly how the points are earned.

Part A. Hybridization of Carbon in H₂CO
The question asks for the hybridization of the carbon atom in H₂CO.
Formaldehyde (H₂CO) has:
- One C=O double bond
- Two C–H single bonds
That gives carbon three regions of electron density. Three regions of electron density correspond to trigonal planar geometry. Trigonal planar geometry means the carbon is sp² hybridized.
Final Answer: sp²
Scoring insight: This part is worth 1 point. The point is earned for clearly stating sp². No additional explanation is required.
Part B. Drawing a Hydrogen Bond
The question asks for a dashed line showing strong hydrogen bonding between one CH₃OH molecule and one H₂CO molecule.
Hydrogen bonding requires:
- A hydrogen attached directly to O, N, or F
- A lone pair on O, N, or F
Methanol (CH₃OH) contains an O–H bond. Formaldehyde (H₂CO) has an oxygen atom with lone pairs but no O–H bond.
So the hydrogen bond must be drawn between:
- The hydrogen attached to oxygen in CH₃OH
- The oxygen atom in H₂CO
The dashed line should connect: H (from O–H in methanol) to O (in formaldehyde)
Important detail: You would lose the point if you drew the dashed line to a hydrogen attached to carbon. C–H bonds do not form hydrogen bonds.
Scoring insight: This part is worth 1 point. The hydrogen bond must be drawn between the correct atoms and in a chemically reasonable orientation.
Part C (i): Choosing a Temperature Where Both Substances Are Liquids
We are given:
- CH₃OH: Melting point is 176 K, Boiling point is 338 K
- H₂CO: Melting point is 181 K, Boiling point is 254 K
For a substance to be liquid, the temperature must be above its melting point and below its boiling point.
For both to be liquids at the same time, the temperature must satisfy both ranges.
- CH₃OH is liquid between 176 K and 338 K
- H₂CO is liquid between 181 K and 254 K
The overlapping liquid range is 181 K to 254 K. Any temperature within that range works.
Example acceptable answer: 200 K
Scoring insight: This earns 1 point for proposing a temperature within the overlapping range.
Part C(ii): Energy Removed During Condensation
The question states that 8.59 g of CH₃OH(l) is present after cooling and asks how much thermal energy was removed to condense it at its boiling point.
We are given:
- Mass of CH₃OH = 8.59 g
- Molar mass = 32.04 g/mol
- ΔHvap = 37.6 kJ/mol
Step 1: Convert grams to moles
- moles = mass ÷ molar mass
- moles = 8.59 g ÷ 32.04 g/mol
- moles ≈ 0.268 mol
Step 2: Multiply by ΔHvap
- Energy = moles × ΔHvap
- Energy = 0.268 mol × 37.6 kJ/mol
- Energy ≈ 10.1 kJ
Because condensation releases heat, the enthalpy change is negative:
ΔH ≈ −10.1 kJ
However, since the question asks for the amount of thermal energy removed, the magnitude 10.1 kJ is typically acceptable.
Scoring insight: This part is worth 1 point for correct mole conversion and energy calculation. Minor rounding differences typically do not prevent full credit if the setup is correct.
How Do You Earn Full Points on AP Chemistry FRQs?
To earn full points on AP Chemistry FRQs, you need more than the correct final answer. The College Board awards points for clear reasoning, correct terminology, and complete justification. Strong responses are structured, precise, and directly aligned with what the question asks.
Focus on the following:
- Show your work clearly and logically
- Use correct scientific terminology
- Justify answers using given data or chemical principles
- Answer exactly what is being asked, nothing less and nothing extra
In AP grading, clarity earns points. Vague statements, missing steps, or unsupported conclusions are where most students lose credit.
Common AP Chem FRQ Mistakes That Cost Easy Points
Let’s be honest. Most point losses are preventable.
Common mistakes include:
- Writing answers without units
- Forgetting significant figures
- Not answering every part of a question
- Giving vague explanations like “more collisions happen”
- Skipping justification in experimental design questions
AP graders do not infer meaning. If you did not clearly write it, you do not earn the point.
How to Practice AP Chemistry FRQs Effectively
If you want to improve quickly, practice with intention.
- Complete timed sets instead of casually solving problems
- Compare your responses with official scoring guidelines
- Rewrite weak explanations until they are precise
- Focus on clarity rather than length
- Practice experimental design questions, since they appear consistently
The fastest way to improve your AP Chemistry FRQ score is to review scoring commentary, not just answer keys. Understanding why points are awarded or lost is what separates average responses from high-scoring ones.
Frequently Asked Questions
AP Chemistry FRQs are scored using detailed rubrics. Each part of a question earns points independently based on demonstrated reasoning, correct terminology, and appropriate justification.
Yes. Points are awarded for individual scoring components. Correct setup, reasoning, or justification may earn credit even if a final calculation contains an error.
The free-response section includes three long-response questions and Four short-response questions. This section accounts for 50 percent of the total exam score.
Long-response questions typically integrate multiple units and are worth 10 points each.
Short-response questions focus on targeted skills and are worth 4 points each, Questions assess conceptual reasoning, calculations, data analysis, and scientific justification
Commonly assessed topics include – Experimental design, Acid–base equilibrium, Thermodynamics and ΔH calculations, Intermolecular forces, Solubility and Ksp, Electrostatics and Coulomb’s law, Graph and data interpretation.
To practice AP Chemistry FRQs effectively, students should work regularly with officially released free-response questions and compare their responses against the official scoring guidelines. Practice should be timed to simulate exam conditions, and weak explanations should be revised for clarity and precision. The goal is not just to reach the correct answer, but to structure responses clearly, use accurate scientific terminology, and fully justify each step according to College Board scoring expectations.
Yes. A calculator is permitted for the entire free-response section.
Disclaimer
This blog is intended for educational and informational purposes only. Any exam-style questions or examples discussed are either adapted from publicly released materials or presented in an original format to illustrate concepts. All explanations reflect own interpretations.
For official exam information, policies, and released materials, please visit the College Board website.



